Research Interest of Dr. Xu Chaohe
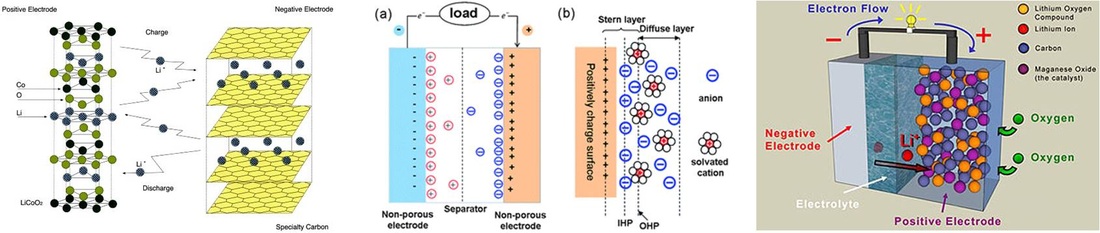
1. Lithium ion batteries (SICCAS)
Lithium-ion batteries are common in consumer electronics. They are one of the most popular types of rechargeable battery for portable electronics, with one of the best energy densities, no memory effect, and only a slow loss of charge when not in use. Beyond consumer electronics, LIBs are also growing in popularity for military, electric vehicle, and aerospace applications. Research is yielding a stream of improvements to traditional LIB technology, focusing on energy density, durability, cost, and intrinsic safety.
My research focused on high energy anode materials, also flexible electrodes for LIBs (based on graphene or carbon nanotubes).
2. Supercapacitors (SICCAS)
Electrical double-layer capacitors (EDLC) are, together with pseudocapacitors, part of a new type of electrochemical capacitors called supercapacitors, also known as ultracapacitors.
Double-layer capacitance – electrostatic storage of the electrical energy achieved by separation of charge in a Helmholtz double layer at the interface between the surface of a conductor electrode and an electrolytic solution electrolyte. The distance of separation of charge in a double-layer is on the order of a few Angstroms (0.3-0.8 nm) and is static in origin.
Pseudocapacitance – Electrochemical storage of the electrical energy, achieved by redox reactions on the surface of the electrode or by specifically adsorpted ions that results in a reversible faradaic charge-transfer on the electrode. The pseudocapacitance is faradaic in origin.
However, the ratio of the two can be very different, depending on the design of the electrodes and the composition of the electrolyte. The pseudocapacitance can increase the capacitance of a supercapacitor by as much as an order of magnitude over that of the double-layer capacitance.
My research focused on graphene based composites as high performance electrodes for Supercapacitor.
3. Metal-air Batteries (NUS)
Lithium–air (Li–air) batteries have recently received much attention due to their extremely high theoretical energy densities. The significantly larger theoretical energy density of the lithium–oxygen batteries is due to the use of a pure lithium metal anode and the fact that the cathode oxidant, oxygen, is stored externally
since it can be readily obtained from the surrounding air. If successfully developed, this battery could provide an energy source for electric vehicles rivaling that of gasoline in terms of usable energy density. However, before Li–air batteries can be realized as high-performance, commercially viable products there are still numerous scientific and technical challenges that must be overcome, from designing the cathode structure, to optimizing the electrolyte compositions and elucidating the complex chemical reactions that occur during charge and discharge.
My research focused on developed bifunctional catalysts for ORR and OER to enhance the electrochemical energy storage.
Lithium-ion batteries are common in consumer electronics. They are one of the most popular types of rechargeable battery for portable electronics, with one of the best energy densities, no memory effect, and only a slow loss of charge when not in use. Beyond consumer electronics, LIBs are also growing in popularity for military, electric vehicle, and aerospace applications. Research is yielding a stream of improvements to traditional LIB technology, focusing on energy density, durability, cost, and intrinsic safety.
My research focused on high energy anode materials, also flexible electrodes for LIBs (based on graphene or carbon nanotubes).
2. Supercapacitors (SICCAS)
Electrical double-layer capacitors (EDLC) are, together with pseudocapacitors, part of a new type of electrochemical capacitors called supercapacitors, also known as ultracapacitors.
Double-layer capacitance – electrostatic storage of the electrical energy achieved by separation of charge in a Helmholtz double layer at the interface between the surface of a conductor electrode and an electrolytic solution electrolyte. The distance of separation of charge in a double-layer is on the order of a few Angstroms (0.3-0.8 nm) and is static in origin.
Pseudocapacitance – Electrochemical storage of the electrical energy, achieved by redox reactions on the surface of the electrode or by specifically adsorpted ions that results in a reversible faradaic charge-transfer on the electrode. The pseudocapacitance is faradaic in origin.
However, the ratio of the two can be very different, depending on the design of the electrodes and the composition of the electrolyte. The pseudocapacitance can increase the capacitance of a supercapacitor by as much as an order of magnitude over that of the double-layer capacitance.
My research focused on graphene based composites as high performance electrodes for Supercapacitor.
3. Metal-air Batteries (NUS)
Lithium–air (Li–air) batteries have recently received much attention due to their extremely high theoretical energy densities. The significantly larger theoretical energy density of the lithium–oxygen batteries is due to the use of a pure lithium metal anode and the fact that the cathode oxidant, oxygen, is stored externally
since it can be readily obtained from the surrounding air. If successfully developed, this battery could provide an energy source for electric vehicles rivaling that of gasoline in terms of usable energy density. However, before Li–air batteries can be realized as high-performance, commercially viable products there are still numerous scientific and technical challenges that must be overcome, from designing the cathode structure, to optimizing the electrolyte compositions and elucidating the complex chemical reactions that occur during charge and discharge.
My research focused on developed bifunctional catalysts for ORR and OER to enhance the electrochemical energy storage.